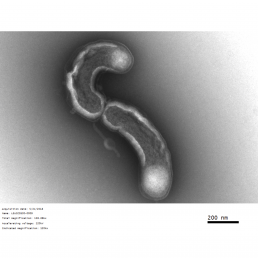
microbiology lab


microbiology lab


microbiology lab


microbiology lab


microbiology lab


microbiology lab


microbiology lab


microbiology lab


microbiology lab

Team
Research
2026
Cheng, Chuankai, Brittany D. Bennett, Pratixa Savalia, Hasti Asrari, Carmen Biel, Kate A. Evans, Rui Tang, and J. Cameron Thrash. 2026. Cell cycle dysregulation of globally important SAR11 bacteria resulting from environmental perturbation. Nature Microbiology. doi: 10.1038/s41564-025-02237-8. On bioRxiv.
Reynolds, Ryan, Anna C.B. Weiss, Chase C. James, Conner Y. Kojima, J.L. Weissman, J. Cameron Thrash, and Naomi M. Levine. 2026. Emergent Metabolic Niches for Marine Heterotrophs. Science Advances. In Press. On bioRxiv.
De Santiago, Alejandro, Shelby J. Barnes, Tiago J. Pereira, Mirayana Marcellino-Barros, Lekeah Durden, Min K. Han, J. Cameron Thrash, and Holly M. Bik. 2025. Pseudoalteromonas is a novel symbiont of marine invertebrates that exhibits broad patterns of phylosymbiosis. Submitted. On bioRxiv.
2025
Coelho, Jordan T., Lauren Teubner, and J. Cameron Thrash. 2025. Detergent-based separation of microbes from marine particles. Applied and Environmental Microbiology 0:e01426-25. doi: 10.1128/aem.01426-25.
Kojima, Conner Y., Michael W. Henson, Jordan T. Coelho, V. Celeste Lanclos, David Bañuelas, and J. Cameron Thrash. 2025. Metagenomes and 1,313 metagenome-assembled genomes from a northern Gulf of Mexico coastal time series. Scientific Data 12, 1388. doi:10.1038/s41597-025-05736-9.
Coelho, Jordan T., Lauren Teubner, Michael W. Henson, V. Celeste Lanclos, Conner Y. Kojima, and J. Cameron Thrash. 2025. Culture-supported ecophysiology of the SAR116 clade demonstrates metabolic and spatial niche partitioning. The ISME Journal 19(1):wraf124. doi:10.1093/ismejo/wraf124
De Santiago, Alejandro, Shelby J. Barnes, Tiago J. Pereira, Mirayana Marcellino-Barros, Holly M. Bik, and J. Cameron Thrash. 2025. Complete Genome Sequences Of Two Pseudoalteromonas undina Strains Isolated From A Marine Nematode (Oncholaimidae) Collected At Tybee Island. Microbiology Resource Announcements. doi:10.1128/mra.00419-25.
Bennett, Brittany D., David A.O. Meier, V. Celeste Lanclos, Hasti Asrari, John D. Coates, and J. Cameron Thrash. 2025. Polyhydroxybutyrate production in freshwater SAR11 (LD12). The ISME Journal 19(1):wraf087. doi: 10.1093/ismejo/wraf087.
Lanclos, V. Celeste, Xiaoyuan Feng, Chuankai Cheng, Mingyu Yang, Cole J. Hider, Jordan T. Coelho, Conner Y. Kojima, Shelby J. Barnes, Catie S. Cleveland, Mei Xie, Yanlin Zhao, Haiwei Luo, and J. Cameron Thrash. 2025. New isolates refine the ecophysiology of the Roseobacter CHAB-I-5 lineage. ISME Communications 5(1):ycaf068. doi:10.1093/ismeco/ycaf068
2024
Henson, Michael W. and J. Cameron Thrash. 2024. Microbial ecology of coastal northern Gulf of Mexico waters. mSystems e01318-23. doi:10.1128/msystems.01318-23.
Layoun, Paul, Mario López-Pérez, Jose M. Haro-Moreno, Markus Haber, J. Cameron Thrash, Michael W. Henson, Vinicius S. Kavagutti, Rohit Ghai, and Michaela M. Salcher. 2024. Microdiversity in genome-streamlined freshwater ‘Ca. Methylopumilus sp.’ and their marine sister lineage OM43. The ISME Journal. 18:1. doi:10.1093/ismejo/wrad036.
2023
Lanclos, V. Celeste, Anna N. Rasmussen, Conner Y. Kojima, Chuankai Cheng, Michael W. Henson, Brant Faircloth, Christopher A. Francis, and J. Cameron Thrash. 2023. Ecophysiology and genomics of the brackish water adapted SAR11 subclade IIIa. The ISME Journal 17: 620-629. doi:10.1038/s41396-023-01376-2.
Getz, Eric W., V. Celeste Lanclos, Conner Y. Kojima, Chuankai Cheng, Michael W. Henson, Max Emil Schön, Thijs J. G. Ettema, Brant C. Faircloth, and J. Cameron Thrash. 2023. The Aegean-169 clade of bacterioplankton is synonymous with SAR11 subclade V (HIMB59) and metabolically distinct. mSystems e00179-23. doi: 10.1128/msystems.00179-23.
Barnes, Shelby J., Raven C. Althouse, Bianca F. Costa, Boyan Hu, Maxim Kovalev, Timur Kulik, Yu-Tung Lee, Meredith C. Moore, Emily Peng, Jing Yao Pook, Akshita Sharma, Celia Wood, Eric A. Webb, Hannah Sterling, Christoph Aeppli, and J. Cameron Thrash. 2023. Metagenome-assembled genomes from photo-oxidized and non-oxidized oil-degrading marine microcosms. Microbiology Resource Announcements. doi:10.1128/mra.00210-23.
Phillips, Elise, Jacob Shaffer, Michael W. Henson, Jordan T. Coelho, Mark O. Martin, and J. Cameron Thrash. 2023. Genome Sequences of Four Agarolytic Bacteria from the Bacteroidia and Gammaproteobacteria. Microbiology Resource Announcements. doi:10.1128/mra.00667-23.
Stapelfeldt, Holly R.D., V. Celeste Lanclos, Michael W. Henson, and J. Cameron Thrash. 2023. Draft genome sequence of the BAL58 Betaproteobacteria representative strain LSUCC0117. Microbiology Resource Announcements. doi:10.1128/MRA.00620-23.
Howe, Kathryn L., Kiley W. Seitz, Lauren G. Campbell, Brett J. Baker, J. Cameron Thrash, Nancy N. Rabalais, Mary-Kate Rogener, Samantha B. Joye, and Olivia U. Mason. 2023. Metagenomics and metatranscriptomics reveal broadly distributed, active, novel methanotrophs in the Gulf of Mexico hypoxic zone and in the marine water column. FEMS Microbiology Ecology. doi: 10.1093/femsec/fiac153.
Kelly, Kyla J., Dieuwertje J. Kast, Cara E. Schiksnis, and J. Cameron Thrash. 2023. Hands-on Hypoxia: Engaging High School Educators in the science behind Marine Microbial Dynamics in Hypoxic Coastal Areas Through Field and Classroom Experiences. Current: The Journal of Marine Education. 38(1): 28–41. doi:10.5334/cjme.86
2022
Kojima, Conner Y., Eric W. Getz, and J. Cameron Thrash. 2022. RRAP: RPKM Recruitment Analysis Pipeline. Microbiology Resource Announcements. doi:10.1128/mra.00644-22.
Lanclos, V. Celeste, Jordan T. Coelho, Catie S. Cleveland, Alex J. Hyer, Mindy M. Brooks, Emily R. Savoie, Scott Kosiba, and J. Cameron Thrash. 2022. A CURE for physiological characterization of bacterioplankton in liquid culture. Journal of Microbiology and Biology Education. doi:10.1128/jmbe.00068-22.
Stapelfeldt, Holly R. D., Shelby J. Barnes, Michael W. Henson, J. Cameron Thrash. 2022. Draft Genome Sequence of the Marine Flavobacteriaceae sp. Strain LSUCC0859. Microbiology Resource Announcements. e00186-22. doi: 10.1128/mra.00186-22.
2021
Savoie, Emily R., V. Celeste Lanclos, Michael W. Henson, Chuankai Cheng, Eric W. Getz, Shelby J. Barnes, Douglas E. LaRowe, Michael S. Rappé, and J. Cameron Thrash. 2021. Ecophysiology of the cosmopolitan OM252 bacterioplankton (Gammaproteobacteria). mSystems 6:e00276-21. doi: 10.1128/mSystems.00276-21.
Xiaoyuan Feng, Xiao Chu, Yang Qian, Michael W. Henson, V. Celeste Lanclos, Fang Qin, Shelby J. Barnes, Yanlin Zhao, J. Cameron Thrash, and Haiwei Luo. 2021. Mechanisms Driving Genome Reduction of a Novel Roseobacter Lineage. The ISME Journal AOP. doi: 10.1038/s41396-021-01036-3.
Thrash, J. Cameron. 2021. Crystal Ball: Towards culturing the microbe of your choice. Environmental Microbiology Reports 13(1): 36-41. doi:10.1111/1758-2229.12898.
Nayfach, Stephen, Simon Roux, Rekha Seshadri, Daniel Udwary, Neha Varghese, Frederik Schulz, Dongying Wu, David Paez-Espino, I-Min Chen, Marcel Huntemann, Krishna Palaniappan, Joshua Ladau, Supratim Mukherjee, T. B. K. Reddy, Torben Nielsen, Edward Kirton, José P. Faria, Janaka N. Edirisinghe, Christopher S. Henry, Sean P. Jungbluth, Dylan Chivian, Paramvir Dehal, Elisha M. Wood-Charlson, Adam P. Arkin, Susannah G. Tringe, Axel Visel, IMG/M Data Consortium☥, Tanja Woyke, Nigel J. Mouncey, Natalia N. Ivanova, Nikos C. Kyrpides & Emiley A. Eloe-Fadrosh. 2021. A genomic catalog of Earth’s microbiomes. Nature Biotechnology 39: 499-509. doi: 10.1038/s41587-020-0718-6. ☥J. Cameron Thrash as Consortium member.
Cheng, Chuankai and J. Cameron Thrash. 2021. sparse-growth-curve: A Computational Pipeline for Parsing Cellular Growth Curves with Low Temporal Resolution. Microbiology Resource Announcements 10:e00296-21. doi: 10.1128/MRA.00296-21
2020
Henson, Michael W., V. Celeste Lanclos, David M. Pitre, Jessica Lee Weckhorst, Anna M. Lucchesi, Chuankai Cheng, Ben Temperton, and J. Cameron Thrash. 2020. Expanding the diversity of bacterioplankton isolates and modeling isolation efficacy with large scale dilution-to-extinction cultivation. Applied and Environmental Microbiology 86:e00943-20. doi: 10.1128/AEM.00943-20
Kujawinski, Elizabeth B., Christopher M. Reddy, Ryan P. Rodgers, J. Cameron Thrash, David L. Valentine, and Helen K. White. 2020. The Deepwater Horizon: Scientific Insights and Advances in Oil Chemistry and Microbiology since 2010. Nature Reviews Earth & Environment. doi:10.1038/s43017-020-0046-x.
-
- Read the WHOI press release
- Read a summary and listen to a related interview with Liz Kujawinski on CBC Radio-Canada
Murray, Alison E., John Freudenstein, Simonetta Gribaldo, Roland Hatzenpichler, Philip Hugenholtz, Peter Kämpfer, Konstantinos T. Konstantinidis, Christopher E. Lane, R. Thane Papke, Donovan H. Parks, Ramon Rossello-Mora, Matthew B. Stott, Iain C. Sutcliffe, J. Cameron Thrash, Stephanus N. Venter, William B. Whitman, Silvia G. Acinas, Rudolf I. Amann, Karthik Anantharaman, Jean Armengaud, Brett J. Baker, Roman A. Barco, Helge B. Bode, Eric S. Boyd, Carrie L. Brady, Paul Carini, Patrick S.G. Chain, Daniel R. Colman, Kristen M. DeAngelis, Maria Asuncion de los Rios, Paulina Estrada de los Santos, Christopher A. Dunlap, Jonathan A. Eisen, David Emerson, Thijs J.G. Ettema, Damien Eveillard, Peter R. Girguis, Ute Hentschel, James T. Hollibaugh, Laura A. Hug, William P. Inskeep, Elena P. Ivanova, Hans-Peter Klenk, Wen-Jun Li, Karen G. Lloyd, Frank E. Löffler , Thulani Makhalanyane, Duane P. Moser, Takuro Nunoura, Marike Palmer, Victor Parro, Carlos Pedrós-Alió, Alexander J. Probst, Theo H.M. Smits, Andrew D. Steen, Emma T. Steenkamp, Anja Spang, Frank J. Stewart, James M. Tiedje, Peter Vandamme, Michael Wagner, Feng-Ping Wang, Brian P. Hedlund, Anna-Louise Reysenbach. 2020. A roadmap for naming the uncultivated majority. Nature Microbiology. doi:10.1038/s41564-020-0733-x
Lucchesi, Anna M., Michael W. Henson, Ben Temperton, and J. Cameron Thrash. 2020. Complete genome of Marinobacterium sp. strain LSUCC0821, isolated from the coastal Gulf of Mexico. Microbiology Resource Announcements 9:e01035-20. doi:10.1128/MRA.01035-20
Yemenian, Tiffany, Kyra M. Florea, J. Cameron Thrash, and Eric A. Webb. 2020. Metagenome-assembled genome of Aphanizomenon flos-aquae strain Clear-A1 from an enrichment culture. Microbiology Resource Announcements 9:e01124-20. doi:10.1128/MRA.01124-20
Florea, Kyra M., J. Cameron Thrash, and Eric A. Webb. 2020. Metagenome-assembled genome of Dolichospermum circinale strain Clear-D4 from a harmful cyanobacterial bloom enrichment culture. Microbiology Resource Announcements 9:e01123-20. doi:10.1128/MRA.01123-20
Floback, Alexis E., Kyra M. Florea, J. Cameron Thrash, and Eric A. Webb. 2020. Vulcanococcus sp. strain Clear-D1 metagenome-assembled genome reconstructed from a cyanobacterial enrichment culture. Microbiology Resource Announcements 9:e01121-20. doi:10.1128/MRA.01121-20
Cheng, Chuankai, Kyra M. Florea, Eric A. Webb, and J. Cameron Thrash. 2020. Metagenome-assembled genome of a Rhodobacteraceae bacterium strain Clear-D3 isolated from a Dolichospermum consortium from Clear Lake, CA. Microbiology Resource Announcements 9:e01119-20. doi:10.1128/MRA.01119-20
Al-Saud, Shaikha, Kyra M. Florea, Eric A. Webb, and J. Cameron Thrash. 2020. Metagenome-assembled genome of Kappabacteriales bacterium strain Clear-D13 from a harmful algal bloom enrichment culture. Microbiology Resource Announcements 9:e01118-20. doi:10.1128/MRA.01118-20
Henson, Michael W., Megan E. Guidry, M. Katherine Carnes, and J. Cameron Thrash. 2020. Draft Genome Sequence of the Novel Coastal Bacterium LSUCC0115 from the MWH-UniPo Clade, Order Burkholderiales, Class Betaproteobacteria. Microbiology Resource Announcements 9:e01492-19. doi:10.1128/MRA.01492-19
Dunning Hotopp, Julie C., David A. Baltrus, Vincent M. Bruno, John J. Dennehy, Steven R. Gill, Julia A. Maresca, Jelle Matthijnssens, Irene L. G. Newton, Catherine Putonti, David A. Rasko, Antonis Rokas, Simon Roux, Jason E. Stajich, Kenneth M. Stedman, Frank J. Stewart, J. Cameron Thrash. 2020. Best Practices for Successfully Writing and Publishing a Genome Announcement in Microbiology Resource Announcements. Microbiology Resource Announcements 9:e00763-20. doi:10.1128/MRA.00763-20
2019
Steen, Andrew D., Alex Crits-Christoph, Paul Carini, Kristen M. DeAngelis, Noah Fierer, Karen G. Lloyd, and J. Cameron Thrash. 2019. High proportions of bacteria and archaea across most biomes remain uncultured. The ISME Journal. AOP doi:10.1038/s41396-019-0484-y
-
- Read the “Behind the Paper” commentary by Drew Steen.
Thrash, J. Cameron. 2019. Culturing the uncultured: risk vs. reward. mSystems 4:e00130-90. doi:10.1128/mSystems.00130-19
Campbell, Lauren G., J. Cameron Thrash, Nancy N. Rabalais, and Olivia U. Mason. 2019. Extent of the annual Gulf of Mexico hypoxic zone influences microbial community structure. PLoS ONE 14(4): e0209055.
Bakshi, Arundhati, Austen T. Webber, Lorelei E. Patrick, E. William Wischusen, and Cameron Thrash. 2019. The CURE for Cultivating Fastidious Microbes. Journal of Microbiology and Biology Education 20(1). doi:10.1128/jmbe.v20i1.1635.
Coelho, Jordan T., Michael W. Henson, and J. Cameron Thrash. 2019. Draft Genome Sequence of Strain LSUCC0112, a Gulf of Mexico Representative of the Oligotrophic Marine Gammaproteobacteria. Microbiology Resource Announcements 8:e00521-19. doi:10.1128/MRA.00521-19
Lanclos, V. Celeste, Michael W. Henson, Chase Doiron, and J. Cameron Thrash. 2019. Draft Genome Sequence of Strain LSUCC0057, a Member of the SAR92 Clade of Gammaproteobacteria. Microbiology Resource Announcements 8:e00599-19. doi:10.1128/MRA.00599-19
2018
Henson, Michael W., V. Celeste Lanclos, Brant C. Faircloth, and J. Cameron Thrash. 2018. Cultivation and genomics of the first freshwater SAR11 (LD12) isolate.The ISME Journal 12:1846-1860. Supplementary Information
Henson, Michael W., Jordan Hanssen, Greg Spooner, Patrick Flemming, Markus Pukonen, Frederick Stahr, and J. Cameron Thrash. 2018. Nutrient dynamics and stream order influence microbial community patterns along a 2914 km transect of the Mississippi River. Limnology and Oceanography 63(5):1837-1855 Supplemental Material
Craine, Joseph, Michael W. Henson, J. Cameron Thrash, Jordan Hanssen, Greg Spooner, Patrick Fleming, Marcus Pukonen, Frederick Stahr, Sarah Spaulding, and Noah Fierer. 2018. Environmental DNA reveals the structure of phytoplankton assemblages along a 2900-km transect in the Mississippi River. bioRxiv preprint.
Thrash, J. Cameron, Brett J. Baker, Kiley W. Seitz, Ben Temperton, Lauren Gillies Campbell, Nancy N. Rabalais, Bernard Henrissat, and Olivia U. Mason. 2018. Metagenomic assembly and prokaryotic MAGs from the northern Gulf of Mexico “Dead Zone”. Microbiology Resource Announcements 7:e01033-18 Table S1
2017
Thrash, J. Cameron , Kiley W. Seitz, Brett J. Baker, Ben Temperton, Lauren E. Gillies, Nancy N. Rabalais, Bernard Henrissat, Olivia U. Mason. 2017. Metabolic roles of uncultivated bacterioplankton lineages in the northern Gulf of Mexico “Dead Zone.” mBio 8(5):e01017-17 Supplementary Information. (Issue Cover)
Halsey, Kimberly H., Stephen J. Giovannoni, Martin Graus, Yanlin Zhao, Zachary Landry, J. Cameron Thrash, and Joost de Gouw. 2017. Biological Cycling of Acetaldehyde by Phytoplankton and Bacterioplankton. Limnology & Oceanography 62(6):2650-2661. doi: 10.1002/lno.10596
2016
Henson, Michael W., David M. Pitre, Jessica Lee Weckhorst, V. Celeste Lanclos, Austen T. Webber, and J. Cameron Thrash. 2016. Artificial seawater media facilitates cultivating members of the microbial majority from the Gulf of Mexico. mSphere 1(2). doi: 10.1128/mSphere.00028-16 Supplementary Information.
Schloss, Patrick, Rene Girard, Thomas Martin, Joshua Edwards, and J. Cameron Thrash. 2016. The status of the archaeal and bacterial census: an update. mBio 7(3):e00201-16
Sun, Jing, Jonathan D. Todd, J. Cameron Thrash, Yanping Qian, Michael C. Qian, Ben Temperton, Jiazhen Guo, Emily K. Fowler, Joshua T. Aldrich, Carrie D. Nicora, Mary S. Lipton, Richard D. Smith, Patrick De Leenheer, Samuel H. Payne, Andrew W. B. Johnston, Cleo Davie-Martin, and Stephen J. Giovannoni. 2016. The abundant marine bacterium Pelagibacter simultaneously catabolizes dimethylsulfoniopropionate to the gases dimethyl sulfide and methanethiol. Nature Microbiology. doi: 10.1038/NMICROBIOL.2016.65
Hogle, Shane L., J. Cameron Thrash, Chris L. Dupont, and Katherine A. Barbeau. 2016. Trace metal acquisition by heterotrophic bacterioplankton with contrasting trophic strategies. Applied and Environmental Microbiology. 82(5): 1613-1624.
Learman, Deric R., Michael W. Henson, J. Cameron Thrash, Ben Temperton, Pamela M. Brannock, Scott R. Santos, Andrew R. Mahon, and Kenneth M. Halanych. 2016. Biogeochemical and microbial variation across 5500 km of Antarctic surface sediment implicates organic matter as a driver of benthic community structure. Frontiers in Microbiology 7:284. doi: 10.3389/fmicb.2016.00284
Lanclos, V. Celeste, Michael W. Henson, David M. Pitre, and J. Cameron Thrash. 2016. Draft Genome Sequence of strain LSUCC0135, an early diverging member of the Methylophilales in the Betaproteobacteria. Genome Announcements. 4(6):e01231-16
2015
Carini, Paul, Benjamin A. S. Van Mooy, J. Cameron Thrash, Angelicque E. White, Yanlin Zhao, Emily O. Campbell, Helen Fredericks, and Stephen J. Giovannoni. 2015. SAR11 lipid renovation in response to phosphorous starvation. PNAS. 112(25): 7677-7772
Gilles, Lauren E., J. Cameron Thrash, Sergio deRada, Nancy N. Rabalais, and Olivia U. Mason. 2015. Archaeal enrichment in the hypoxic zone in the northern Gulf of Mexico. Environmental Microbiology 17(10): 3847-3856
Thrash, J. Cameron, Jessica Lee Weckhorst and David M. Pitre. 2015. Cultivating Fastidious Microbes. In Hydrocarbon and Lipid Microbiology Protocols, vol. 4 (Cultivation). Edited by Terry J. McGenity, Kenneth N. Timmis and Balbina Nogales.
2014
Thrash, J. Cameron, Ben Temperton, Brandon K. Swan, Zachary C. Landry, Tanja Woyke, Edward F. DeLong, Ramunas Stepanauskas, and Stephen J. Giovannoni. 2014. Single-cell enabled comparative genomics of a deep ocean SAR11 bathytype. The ISME Journal 8(7): 1440-1451
Giovannoni, Stephen J., J. Cameron Thrash, and Ben Temperton. 2014. Implications of streamlining theory for microbial ecology. The ISME Journal 8(8): 1553-1565.
Carini, Paul, Emily O. Campbell, Jeff Morré, Sergio Sañudo-Wilhelmy, J. Cameron Thrash, Samuel Bennett, Ben Temperton, Tadhg Begley, and Stephen J. Giovannoni. 2014. Discovery of a SAR11 growth requirement for thiamin’s pyrimidine precursor and its distribution in the Sargasso Sea. The ISME Journal 8(8): 1727-1738
2013
Ferla, Matteo P., J. Cameron Thrash, Stephen J. Giovannoni, and Wayne M. Patrick. 2013. New rRNA Gene-Based Phylogenies of the Alphaproteobacteria Provide Perspective on Major Groups, Mitochondrial Ancestry and Phylogenetic Instability. PLOS ONE 8(12): e83383. doi:10.1371/journal.pone.0083383
Smith, Daniel P., J. Cameron Thrash, Carrie D. Nicora, Mary S. Lipton, Kristin E. Burnum, Paul Carini, Richard D. Smith, and Stephen J. Giovannoni. 2013. Proteomic and transcriptomic analysis of “Candidatus Pelagibacter ubique” describes the first PII-independent response to nitrogen limitation in a free-living alphaproteobacterium. mBio 4(6):e00133-12. doi:10.1128/mBio.00133-12
Vergin, Kevin L.*, Bánk Beszteri*, Adam Monier, J. Cameron Thrash, Ben Temperton, Alexander H. Treusch, Fabian Kilpert, Alexandra Z. Worden, and Stephen J. Giovannoni. 2013. High resolution SAR11 ecotype dynamics at the Bermuda Atlantic Time-series Study Site by phylogenetic placement of pyrosequences. The ISME Journal 7(7): 1322-1332
Zhao, Yanlin*, Ben Temperton*, J. Cameron Thrash, Michael S. Schwalbach, Kevin L. Vergin, Zachary C. Landry, Mark Ellisman, Tom Deerinck, Matthew B. Sullivan, and Stephen J. Giovannoni. 2013. Abundant SAR11 viruses in the ocean. Nature 494(7437): 357-360
2012
Grote, Jana*, J. Cameron Thrash*, Megan J. Huggett, Zachary C. Landry, Paul Carini, Stephen J. Giovannoni, and Michael S. Rappé. 2012. Streamlining and Core Genome Conservation among Highly Divergent Members of the SAR11 Clade. mBio 3(5): e00252-12
2011
Thrash, J. Cameron, Alex Boyd, Megan J. Huggett, Jana Grote, Paul Carini, Ryan J. Yoder, Barbara Robbertse, Joseph W. Spatafora, Michael S. Rappé, and Stephen J. Giovannoni. 2011. Phylogenomic evidence for a common ancestor of mitochondria and the SAR11 clade. Scientific Reports 1(13); DOI:10.1038/srep00013
Sun, Jing, Laura Steindler, J. Cameron Thrash, Kimberly H. Halsey, Daniel P. Smith, Amy E. Carter, Zachary C. Landry, and Stephen J. Giovannoni. 2011. One Carbon Metabolism in SAR11 Pelagic Marine Bacteria. PLOS ONE 6(8): e23973
Dreher, Theo W., Nathan Brown, Connie S. Bozarth, Andrew D. Schwartz, Erin Riscoe, Cameron Thrash, Samuel E. Bennett, Shin-Cheng Tzeng, and Claudia S. Maier. 2011. A freshwater cyanophage whose genome indicates close relationships to photosynthetic marine cyanomyophages. Environmental Microbiology 13(7): 1858-1874
Wrighton, Kelly C., J. Cameron Thrash, Ryan A. Melnyk, J. P. Bigi, Katherine G. Byrne-Bailey, J. P. Remis, Denise Schichnes, M. Auer, C. J. Chang, and John D. Coates. 2011. Evidence for direct electron transfer by a Gram-positive bacterium isolated from a microbial fuel cell. Applied and Environmental Microbiology 77(21): 7633-7639
Weber, Karrie A., J. Cameron Thrash, J. Ian Van Trump, Laurie A. Achenbach, and John D. Coates. 2011. Environmental and Taxonomic Bacterial Diversity of Anaerobic Uranium(IV) Bio-Oxidation. Applied and Environmental Microbiology 77(13): 4693-4696
Van Trump, J. Ian, Kelly C. Wrighton, J. Cameron Thrash, Karrie A. Weber, Gary L. Andersen, and John D. Coates. 2011. Humic acid-oxidizing, nitrate-reducing bacteria in agricultural soils. mBio 2(4); DOI: 10.1128/mBio.00044-11
Thrash, J. Cameron*, Jang-Cheon Cho*, Anthony D. Bertagnolli, Steve Ferriera, Justin Johnson, Kevin L. Vergin, and Stephen J. Giovannoni. 2011. Genome sequence of the marine Janibacter sp. strain HTCC2649. Journal of Bacteriology 193(2): 584-585
Thrash, J. Cameron and John D. Coates. 2011. Phylum XVII. Acidobacteria phy. nov. In Bergey’s Manual of Systematic Bacteriology, 2nd edn, vol. 4 (The Bacteroidetes, Planctomycetes, Chlamydiae, Spirochaetes, Fibrobacteres, Fusobacteria, Acidobacteria, Verrucomicrobia, Dictyoglomi and Gemmatimonadetes). Edited by Noel R. Krieg, James T. Staley, Brian Hedlund, Bruce J. Paster, Naomi Ward, Wolfgang Ludwig, and William B. Whitman.
Thrash, J. Cameron, Sarir Ahmadi, and John D. Coates. 2011. Anaerobic respiratory iron(II) oxidation. In Microbial Metal and Metalloid Metabolism: Advances and Applications. Edited by John F. Stolz and Ronald S. Oremland
2010
Thrash, J. Cameron, Sarir Ahmadi, Tamas Torok, and John D. Coates. 2010. Characterization of Magnetospirillum bellicus sp. nov., a novel dissimilatory perchlorate-reducing bacterium in the Alphaproteobacteria isolated from a bioelectrical reactor. Applied and Environmental Microbiology 76(14): 4730-4737
Thrash, J. Cameron, Jarrod Pollock, Tamas Torok, and John D. Coates. 2010. Description of the novel perchlorate-reducing bacteria Dechlorobacter hydrogenophilus gen. nov., sp. nov., and Propionivibrio militaris, sp. nov. Applied Microbiology and Biotechnology 86(1): 335-343
Melchior, Benoit, Angie E. Garcia, Bor-Kai Hsiung, Katherine M. Lo, Jonathan M. Doose, J. Cameron Thrash, Anna Stalder, Matthias Staufenbiel, Harald Neumann, and Monica J. Carson. 2010. Dual Induction of TREM2 and Tolerance related transcript, Tmem176b in amyloid transgenic mice: implications for vaccine-based therapies for Alzheimer’s disease. ASN NEURO 2(3): 157-170
Thrash, J. Cameron*, Jang-Cheon Cho*, Steve Ferriera, Justin Johnson, Kevin L. Vergin, and Stephen J. Giovannoni. 2010. Genome sequences of Pelagibaca bermudensis HTCC2601T and Maritimibacter alkaliphilus HTCC2654T, the type strains of two marine Roseobacter genera. Journal of Bacteriology 192(20): 5552-5553
Thrash, J. Cameron*, Jang-Cheon Cho*, Steve Ferriera, Justin Johnson, Kevin L. Vergin, and Stephen J. Giovannoni. 2010. Genome sequences of strains HTCC2148 and HTCC2080 belonging to the OM60/NOR5 clade of the Gammaproteobacteria. Journal of Bacteriology 192(14): 3842-3843
Thrash, J. Cameron, Ulrich Stingl, Jang-Cheon Cho, Steve Ferriera, Justin Johnson, Kevin L. Vergin, and Stephen J. Giovannoni. 2010. Genome sequence of the novel marine Gammaproteobacterium strain HTCC5015. Journal of Bacteriology 192(14): 3838-3839
Thrash, J. Cameron*, Jang-Cheon Cho*, Kevin L. Vergin, and Stephen J. Giovannoni. 2010. Genome sequences of Oceanicola granulosus HTCC2516T and O. batsensis HTCC2597T. Journal of Bacteriology 192(13): 3549-3550
Thrash, J. Cameron*, Jang-Cheon Cho*, Kevin L. Vergin, Robert M. Morris, and Stephen J. Giovannoni. 2010. Genome Sequence of Lentisphaera araneosa HTCC2155T, the type species of the order Lentisphaerales in the phylum Lentisphaerae. Journal of Bacteriology 192(11): 2938-2939
2009
Weber, Karrie A., David B. Hedrick, Aaron D. Peacock, J. Cameron Thrash, David C. White, Laurie A. Achenbach, and John D. Coates. 2009. Physiological and taxonomic description of the novel autotrophic, metal oxidizing bacterium, Pseudogulbenkiania sp. strain 2002. Applied Microbiology and Biotechnology 83(3): 555-565
Thrash, J. Cameron, Bruce E. Torbett, and Monica J. Carson. 2009. Developmental Regulation of TREM2 and DAP12 Expression in the Murine CNS: Implications for Nasu-Hakola Disease. Neurochemical Research 34(1): 38-45
2008
Thrash, J. Cameron and John D. Coates. 2008. Review: Direct and Indirect Electrical Stimulation of Microbial Metabolism. Environmental Science and Technology 42(11): 3921-3931
2007
Thrash, J. Cameron, J. Ian Van Trump, Karrie A. Weber, Elisabeth Miller, Laurie A. Achenbach, and John D. Coates. 2007. Electrochemical Stimulation of Microbial Perchlorate Reduction. Environmental Science and Technology 41(5): 1740-1746
Papenfuss, Tracey L., J. Cameron Thrash, Patricia E. Danielson, Pamela E. Foye, Brian S. Hillbrush, Gregor J. Sutcliffe, Caroline C. Whitacre, and Monica J. Carson. 2007. Induction of Golli-MBP Expression in CNS Macrophages During Acute LPS-Induced CNS Inflammation and Experimental Autoimmune Encephalomyelitis (EAE). TheScientificWorldJOURNAL 7(S2): 112-120
Plant, Sheila R., Heather A. Iocca, Ying Wang, J. Cameron Thrash, Brian P. O’Connor, Heather A. Arnett, Yang-Xin Fu, Monica J. Carson, and Jenny P.-Y. Ting. 2007. Lymphotoxin beta receptor (Lt betaR): dual roles in demyelination and remyelination and successful theraputic intervention using Lt betaR-Ig protein. Journal of Neuroscience 27(28): 7429-37
2006
Carson, Monica J., J. Cameron Thrash, and Barbara Walter. 2006. The cellular response in neuroinflammation: The role of leukocytes, microglia and astrocytes in neuronal death and survival. Clinical Neuroscience Research 6(5): 237-245
Plant, Sheila R., Ying Wang, Sophie Vasseur, J. Cameron Thrash, Eileen J. McMahon, Daniel T. Bergstralh, Heather A. Arnett, Steven D. Miller, Monica J. Carson, Juan L. Iovanna, and Jenny P.-Y. Ting. 2006. Upregulation of the stress-associated gene p8 in mouse models of demyelination and in multiple sclerosis tissues. Glia 53(5): 529-37
2004
Carson, Monica J., J. Cameron Thrash, and David Lo. 2004. Analysis of microglial gene expression: identifying targets for CNS neurodegenerative and autoimmune disease. American Journal of Pharmacogenomics 4(5): 321-30
Opportunities
We’ve compiled a list of funding opportunities for early career scientists at multiple levels. If you have interest in joining us, please review the Thrash Lab personnel expectations to see if our culture and attitude look like a fit. If so, email Cameron your CV with some comments about your motivation to work with us and your particular project interests, as well as to enquire about whether we have open positions. Prospective graduate students can apply to join the Thrash lab through the Ph.D. program in Marine and Environmental Microbiology. Independently of open positions, we’re also always open to prospective postdocs who wish to apply for funding through the NSF Postdoctoral Fellowships in Biology, NSF Ocean Science Postdoctoral Research Fellowships, and Simons Postdoctoral Fellowships in Marine Microbial Ecology.
Improving your graduate school expectations
Pursuing your graduate degree is an excellent investment in your future. It’s also important to have appropriate expectations about what graduate school will entail and the possible career outcomes for PhDs in biological sciences. We have specific personnel expectations for the Thrash Lab, and JCT is always willing to discuss these further. Here are some additional resources from other places for information on what to expect during and after graduate school:
Nature’s 2019 survey of over 6,000 Ph.D. students from around the world.
A message for mentors from dissatisfied graduate students.
Infographic: Where will a biology Ph.D. take you?
Ten simple rules for developing good reading habits during graduate school and beyond.
An open letter to graduate students and other procrastinators: it’s time to write
Tips for interacting with your advisor
The care and maintenance of your adviser
Managing up: how to communicate effectively with your PhD adviser
Funding for Undergraduates
- ASM Undergraduate Research Fellowship
- ASM Undergraduate Research Capstone Program
- Barry Goldwater Scholarship
- DoD SMART Scholarship
- Ford Foundation Predoctoral Fellowship
- Howard Hughes Medical Institute
- NASA Undergraduate Student funding opportunities
- NOAA Student Funding Opportunities
- USC Provost’s Undergrad Research Fellowships
- USC Student Opportunities for Academic Research (SOAR)
- USC Summer Undergraduate Research Fund (SURF)
Current and prospective Undergraduates should read this piece in Nature: From lectures to the lab: three steps to becoming an undergraduate researcher
Funding for Graduate Students
The Johns Hopkins List of Graduate Student Funding Opportunities
The Johns Hopkins List of Underrepresented Minority and Low-income Graduate Student Funding Opportunities
- American Association of University Women
- Academy of Underwater Arts & Sciences Zale Parry Scholarship
- ASM Graduate Research Capstone Fellowship
- Chateaubriand Fellowship (10 months of study in France for US students)
- DoD SMART Scholarship
- DoE Computational Science Graduate Fellowship
- DoE Office of Science Graduate Student Research Program
- Ford Foundation Dissertation Fellowship
- Fulbright Fellowship (one year of funding in the US for UK students)
- Hertz Foundation Fellowship
- Howard Hughes Medical Institute
- NASA Graduate Student funding opportunities
- National Defense Science and Engineering Graduate Fellowship
- NOAA Dr. Nancy Foster Scholarship Program
- NSF Graduate Research Fellowship Program (See this excellent help page)
- Sea Grant Knauss Fellowship (1 year policy fellowship in Washington, D.C.)
- USC Graduate Fellowships
Funding for Postdocs
The Johns Hopkins List of Postdoctoral Funding Opportunities
- Agouron Institute Geobiology Fellowships
- American Association of University Women
- ASM Career Development Grants for Postdoctoral Women
- David H. Smith Conservation Research Fellowship
- Ford Foundation Postdoctoral Fellowship
- Human Frontier Science Program Fellowship
- Life Sciences Research Foundation
- L’Oréal USA For Women in Science
- Marie Skłodowska-Curie Individual Fellowships
- NASA Postdoctoral funding opportunities
- NIH Intramural Research Program
- NIH K-Awards for Career Development
- NOAA Climate and Global Change Postdoctoral Fellowships
- NSF Postdoctoral Research Fellowships in Biology
- NSF Postdoctoral Research Fellowship in Ocean Sciences
- Simons Foundation Postdoctoral Fellowships in Marine Microbial Ecology
Education
BISC 300L Introduction to Microbiology
Introduction to Microbiology explores the world of microorganisms via a comparative approach to Bacteria, Archaea, Protists, and Viruses: their structure, life cycles, geochemical activity, ecology, and nutrition. Fundamentals of metabolism and microbial genetics will also be explored alongside evolution and systematics. Co-taught with Dr. Julia Schwartzman.
BISC 473L Biological Oceanography
This course will cover the basics of biological, physical, and chemical dynamics in the oceans with a particular emphasis on life in different ocean environments. Specific topics include primary production of phytoplankton, secondary production by zooplankton, bacterial remineralization, physiology and ecology of fishes, and marine mammals. Co-taught with Dr. Naomi Levine.
BISC 588L Introductory Bioinformatics
Contemporary research in the biological sciences has capitalized on recent advances in computational methodologies to address novel questions relating to the ecology and evolution of life on this planet. To effectively contribute to this growing field, the next generation of biologists must have a solid foundation in both classical biology and bioinformatics. This hands-on course will introduce biologists with minimal to no prior command line experience to basic bioinformatics skills, software, and analysis pipelines. Co-taught with Dr. Carly Kenkel
Microbiology-focused Course-based Undergraduate Research Experience (mCURE) sections
In coordination with Linda Bazilian and Gorjana Bezmalinovic, we are conducting mCURE sections in freshman biology laboratory courses. Students learn theory from primary literature, conduct real experiments to cultivate and characterize microorganisms from marine sources, and learn DNA extraction, PCR, and basic BLAST to identify their isolates. We have two semesters of curricula published: The CURE for Cultivating Fastidious Microbes and A CURE for Physiological Characterization of Bacterioplankton in Liquid Culture.
MicroSeminar
Be sure to check out MicroSeminar (@MicroSeminar), a set of free online microbiology seminars, where Cameron is a co-host with Lizzy Wilbanks, David Baltrus, Jillian Petersen, Sarah Preheim, Laetitia Wilkins (and formerly Jennifer Biddle).
Alums
- Remedios Abrica – USC Undergraduate Researcher 2025
- Dr. David C. Bañuelas – USC Presidential Sustainability Solutions Postdoctoral Fellow (co-advised with Dr. Felipe de Barros). After: Assistant Professor at University of Redlands
- Shelby J. Barnes – USC Research Lab Technician 2020-2024
- Dr. Brittany D. Bennett – USC Postdoctoral Scholar 2020-2024. After: Research Professional at University of Minnesota
- Carmen Biel – USC Undergraduate Researcher 2024
- Brette Campbell – LSU Undergraduate Researcher; B.S. Microbiology, 2014
- Mary Carnes – LSU Undergraduate Researcher 2017-2018
- Rachel Clifford – USC Ph.D. student 2020-2021. After: graduate school at San Jose State Moss Landing Marine Labs
- Maia Dau – USC Undergraduate Researcher 2022-2023
- Chase Doiron – LSU Undergraduate Researcher 2015-2016
- Bailey Dixon – LSU Undergraduate Researcher 2015-2017
- Joshua Edwards – LSU Undergraduate Researcher 2015
- Logan Forest – LSU Undergraduate Researcher 2018
- Eric Getz – USC M.S. student 2020-2022; M.S. Biological Sciences 2022
- Rene Girard – LSU Undergraduate Researcher; B.S. Biology, 2015. After: graduate school at University of Texas Northeast
- Megan Guidry – LSU Undergraduate Researcher 2017-2018
- Dr. Michael W. Henson – LSU Ph.D. student 2014-2018; Ph.D. Biology 2018; USC Postdoc 2019. After: Postdoc with Dr. Maureen Coleman, U of Chicago
- Cole Hider – USC Undergraduate Researcher 2019-2023. After: Medical school at USC
- Alex Hyer – USC Ph.D. student 2018-2019
- Irene Kim – USC Undergraduate Researcher 2025
- Conner Kojima – USC Undergraduate Researcher 2021-2024. After: Medical school at USC
- Margaret Kosierowski – USC Undergraduate Researcher 2023-2025
- Dr. V. Celeste Lanclos – LSU Undergraduate Researcher 2015-2017; LSU graduate student 2018-2019; USC Ph.D. student 2019-2023; Ph.D. Biology 2023. Trusten Award winner 2023. After: Postdoc with Dr. John Coates, UC Berkeley
- Anna Lucchesi – LSU Undergraduate Researcher 2016-2018
- Thomas Martin – LSU Undergraduate Researcher 2015
- Meha Magesh – USC Undergraduate Researcher 2019-2020
- Zainab Neemuchwala – USC Undergraduate Researcher 2023-2025.
- David Pitre – LSU Undergraduate Researcher 2014-2015; B.S. Biology, Amborski Award winner, 2015. After: Medical school at LSU New Orleans
- Arnav Rashid – USC Undergraduate Researcher 2020-2022
- Dr. Nicole R. Ratib – Simons Foundation Postdoctoral Fellow in Marine Microbial Ecology 2020-2022. After: Pivot Bio
- Emily Savoie – LSU Undergraduate Researcher 2014-2018; B.S. Biology, B.S. Microbiology, Amborski Award winner, 2018. After: lab tech with Dr. Jennifer Brum, LSU
- Nicolas Spatafora – LSU Undergraduate Researcher 2014-2015; transferred to Oregon State University
- Deesha Sharma – USC Undergraduate Researcher 2024-2025
- Holly R. D. Stapelfeldt – USC Research Laboratory Technician 2021-2022
- Lauren Teubner – USC Undergraduate Researcher 2021-2024. After: Medical school at the Mayo Clinic
- Lindsay Thomas – USC Undergraduate Researcher 2024-2025
- Mikaela Walters – LSU Undergraduate Researcher 2017-2018
- Austen Webber – LSU Ph.D. student 2015-2016. After: brewer at Mantra Artisan Ales
- Jessica Weckhorst – LSU Undergraduate Researcher 2013-2015; B.S. Microbiology, Socolofski Award winner, 2015. After: Ph.D. from Baylor University
- Celia Wood – USC Undergraduate Researcher 2023-2025.
- Dr. Irene Zhang – USC Postdoctoral Scholar 2023-2025
Contact
University of Southern California
Department of Biological Sciences
3616 Trousdale Pkwy, AHF 209
Los Angeles, CA 90089
thrash at usc dot edu